 Affidavit of Testimony
Affidavit of Testimony
Return to the Cognitive Enhancement Research Institute
Home Page, or Parkinson's
Page.
 Affidavit of Testimony
Affidavit of Testimony------------------------------------------------------------------ Steven Wm. Fowkes (fowkes@ceri.com) Phone: 650-321-CERI Cognitive Enhancement Research Institute Fax: 650-323-3864 P.O.Box 4029, Menlo Park, California, 94026 USA Alt: 650-321-6670 ---------------------http://www.ceri.com/-------------------------
This affidavit consists of a body of text followed by a series of footnotes. Clicking on the footnote number in the main text will jump you to the appropriate footnote. To go back to the text, use the “back” button on your web browser. Some footnotes are cited in other footnotes, so, on occassion, you might have to click “back” twice to return to the main body of text.
This affadavit also contains seven pages of color graphics (click here for list). These have been scanned at half resolution to make them displayable on low-resolution monitors. Several of these may exceed the width of your screen, which will require scrolling to the right to view the entire image.
Two of the exhibits (Exhibit #2 and #4) consist of four panels each. These are broken down into four separate graphic files to make them easier to see, and to place them closer to the place in the text where they are discussed. However, the crowding of images can cause problems when they overlap, which can cause the images to overlap text. If this happens, increase the size of the font used by your web browser software to display text. This should separate the graphics and eliminate the problem.
I am the Executive Director of the Cognitive Enhancement Research Institute, and have been since 1992. At the request of James Kimball, I appeared as an expert defense witness in the Kimball case earlier this year. Mr. Kimball provided for my transportation and lodging; I donated my time and expertise.
Mr. Kimball asked me to testify because of my unique knowledge of deprenyl chemistry and deprenyl products and because he knows that I have an ability to convey technical issues to lay people. In fact, one of my primary job responsibilities at CERI is to write, edit and publish our newsletter, Smart Life News, which explains our analyses of the emerging scientific and medical state of the art in cognitive therapies to both lay and professional audiences. We are able to do this without compromising the essential scientific accuracy of our “translations.” We regularly receive superlative evaluations from our doctor (Ph.D.) and physician (M.D.) subscribers.
CERI operates as a think tank and information clearinghouse for pharmaceutical, herbal and nutritional therapies for cognitive conditions of all types. We deal with overt conditions affecting children (autism, Down’s syndrome, attention deficit/hyperactivity disorders, dyslexia and learning disorders), the elderly (Alzheimer’s disease, Parkinson’s disease, stroke, dementias and senility syndromes), and we deal with subtle cognitive conditions affecting ordinary, healthy, aging people (for examples, metabolic disorders, biorhythm disturbances and hormone deficiencies). We are the only institute of our kind in the world. We spend the majority of our time and resources investigating generic (non-proprietary) technologies.
My extreme investment in the accuracy of my scientific explanations became a problem during my testimony before your court in the Kimball case. During the first set of questions by defense attorney Wardell, a highly technical matter was raised, which should not have been raised at that point during my examination. I had previously discussed this strategic matter with Mr. Wardell (the evening before) and had warned him to bring up the matter later in testimony, after extensive groundwork had been laid so that you and the jury could easily understand the technical distinctions that it was my responsibility to make clear. Due to Mr. Wardell’s unfortunate choice of initial question, this matter was raised without proper context. Looking back, I do not know whether Mr. Wardell 1) made a strategic mistake, 2) misunderstood a technical aspect of my testimony himself, or 3) inadvertently chose the wrong word in developing my testimony. Whatever the reason, a serious travesty of justice has resulted.
It is my opinion that Mr. Wardell intended to ask me about my knowledge of citric acid (also known as citrate), selegiline (also called deprenyl), glycerine (also called glycerol) and water. I am quite familiar with each of those substances. However, his question asked about “all” of the “ingredients” in DEDI’s liquid deprenyl citrate product (LDC). What the word ingredient means to you and the jury (a substance added to a recipe or formula) is not necessarily what it means to a scientist (a substance contained within a sample or product). More importantly, his use of the word “all” made the question absolute.
There are few substances, recipes, formulas or products on the planet for which scientists know each and every chemical entity that they contain. This may seem shocking, but it is true. Such is the chemical reality of the real world. As analytical techniques have improved with advancing technology, more and more substances are being found in increasingly smaller amounts.[1] These newly discovered substances have always been present in the products. We just didn’t know it at the time because of the technological limitations of the then-state-of-the-art testing equipment. The average plant substance (pick pretty much any vegetable or herb) contains approximately 10,000 different chemicals by today’s analytical methods. Most of these remain unidentified.
Given these facts, I was compelled to answer Mr. Warden’s question in the negative. I had taken an oath to tell “the whole truth,” which included not only the primary ingredients (citrate, deprenyl, glycerol and water) but the impurity ingredients as well. Some of these impurities have been characterized, but some have not. Since some of these “ingredients” are uncharacterized, I could not honestly testify that I knew “all” of them. It is my expert opinion that no scientist could have honestly answered that question in the affirmative.
In hindsight, I now regret answering the question within the context of “the whole truth.” I now wish that I had instead answered the question in a non-absolute lay context only — with an added qualifier about the “technical limitation” or “possible misleadingness” of my answer. I could have done this within my oath. One reason I did not was that I was distracted by thoughts about later aspects of my testimony, which the question addressed prematurely. In a sense, I was already ahead of myself. For this, I apologize.
Another reason is that I was nervous. Although I have testified in court on several occasions, I do not have the extensive experience that would be necessary for me to be calm enough to anticipate the possible ways in which you might have misunderstood my answer, and calm enough to handle the emotional tone of your reaction. I could see that there was a problem, but I didn’t really have a clue as to what would prompt you to say what you said to me and the court. When Mr. Warder failed to ask you for an opportunity for me to clarify my response, so that the matter to which you were reacting could have been made the specific content of my testimony, I did make one attempt to intercede with you directly, which I considered my responsibility, despite the intense intimidation I was feeling from your behavior.
In the interests of justice, I am providing this affidavit now, to make clear what I would have testified to then, if I had been allowed the opportunity to “tell the whole truth” to the best of my ability.
What are “ingredients”? A common lay misperception is that the ingredients on the label are the ingredients in a product. In reality, products may contain many substances that are not on the label. These may be ingredients that the FDA does not require to be labeled (such as BHT food preservative added by a previous manufacturer of a fat-containing ingredient, like lard, vegetable oil or peanut butter which is listed on the label). Ingredients may also be impurities, reaction byproducts, or degradation products associated with ingredients in the product — or produced by biological organisms in the product.
Each of the primary ingredients in LDC (even water) contains impurities that can be detected by scientists. These become part of the final LDC product.[2] I considered all of these to be encompassed by Mr. Warder’s question.
Ingredients may also be contaminants from packaging materials, that leech into the product during storage. For example, bottled water contains an estrogenic chemical (bisphenol A) which has leached out of the FDA-approved polycarbonate plastic water bottles in which the water is packaged. Although the FDA considers the levels low enough to be acceptably safe, a scientist must acknowledge that they are present in the bottled water that we drink from such containers. I presume that it would have been my responsibility as an expert witness to discuss such trace impurities in any court case dealing with possible harm (like cancer) from drinking such bottled water.
I came to your court prepared to discuss the role of a variety of impurities that are found in deprenyl products. These impurities have a direct bearing on whether there are or are not differences between different deprenyl products regarding their safety, efficacy, side effects, and identity. Since I was prevented from discussing these issues in court, I will take the opportunity to discuss them now.
One of the technical aspects of chemistry, which I tried to relate you, is that impurities are often unknown. In other words, scientists know that impurities are present long before they figure out what those impurities are. Chemists can even quantify the amount of each impurity without knowing in any way what they are. They can even quantify a group of impurities, without knowing how many are present. Most of the dietary supplements, over-the-counter medications and FDA-approved drugs contain impurities which are presently unknown. For economic and technical reasons, this state of affairs is likely to stay that way for some time. In some commercial products (i.e., foods and herbs), a majority of the substances known to be present remain uncharacterized at a molecular (chemical) level.
Scientists must also acknowledge that there may be substances that are present in amounts too small to be detected.[1] As analytical devices and techniques have improved, the number of unknowns has increased. There is no reason to think that this will not continue into the future.
The prevalence and variety of impurities in substances makes each substance in the world fundamentally unique. For practical and safety reasons, we institutionalize this uniqueness in laws, regulations rules and traditions. For example, Bayer aspirin is different from generic aspirins by legal criteria, regulatory criteria and chemical criteria. This is why the FDA requires independent approval for each brand of product.
As another example, extracts of St. John’s wort are different between batches 1) harvested in different years, 2) grown in differing climates, 3) harvested at different times, 4) grown at different altitudes, and 5) which have and have not been subjected to insect predation. This is why some companies “standardize” their herbal extracts to one or more specific compounds in the extract which are considered to be “active” ingredients.[3]
Even batches of synthetic drugs are different from each other. That is why government regulators require pharmaceutical companies to use batch and lot numbers as part of good manufacturing practice. If the uniqueness of a particular batch is later found to be a problem, the offending lots (and only the offending lots) can be efficiently recalled.
The fact that an impurity is unknown is not considered a reasonable basis for assuming any degree of risk. Yet we know that tiny trace impurities can be the cause of severe consequences. Take the case of Showa Denko’s L-tryptophan, an essential (dietarily required) amino acid sold as a dietary supplement for more than three decades. Showa Denko’s tryptophan met every FDA standard for purity and safety, yet an impurity of far less than a tenth of one percent killed dozens of US citizens. Such toxic unknowns are described as “peak E” or “peak X” based on the sequence of “blips” that occur on a sophisticated analytical device called a chromatograph.[4] However, there are far more peaks that are “safe” (not case associated) than there are “toxic” (case-associated). Determinations of which peaks are associated with adverse effects in people and which are not is the chemical or epidemiological equivalent of “guilt by association.” It takes additional scientific investigation to establish a causal relationship between the suspected “unknown” and the disease being investigated. Knowledge of the chemical identity of the unknown substance is completely unnecessary to the task of establishing a cause-and-effect relationship to a disease process.
Scientific expertise must be based on scientific principles and methodologies. Scientific principles and methodologies establish the ways in which one may conclude causal or associational relationships between observations, events and facts (data). Because scientists are human, they can make mistakes. They can also be motivated by economic and political ends which can undermine the scientific process. This is why we closely examine 1) the study design, 2) the methodologies used to acquire data, 3) the statistical methods used to analyze the data, and 4) the reasoning of the conclusion. Often, there are flaws. It is my job to analyze scientific reports and the data they contain for weaknesses. In performing that job, I discovered an egregious mistake in a major scientific paper on deprenyl (specifically Eldepryl-brand deprenyl).[5] When correctly analyzed, this paper provides direct evidence that the impurities in Eldepryl were of life-threatening consequence to the Parkinson’s patients enrolled in that study. This is of extreme relevance to the Kimball case.
I have been collecting reports (professional and patient anecdotes) on DEDI’s liquid deprenyl citrate for more than eight years. Initially, I assumed, as do most medical professionals, that it could have only subtle advantages over Eldepryl. Most differences between brand-name and generic products are subtle. Some physicians, governmental bureaucrats, insurance companies and politicians even claim that there are no significant clinical differences between generics and name brand pruducts except price. Other physicians dispute such presumptions.
Nevertheless, within a strictly scientific context, differences between different brands of products exist. These differences can be clearly seen in impurity profiles derived from high pressure liquid chromatography (HPLC) or gas chromatography (GC). We can even identify batch-to-batch differences between different lots of a specific branded products. The scientific issue is not whether such differences exist, but whether or not they are important. Are any of the peaks pharmacologically active? Do they affect risk? Do they increase or decrease product efficacy? Without specific data, answers to these questions are rank speculation.
At the same time that I was receiving reports of LDC, I have been studying the scientific/medical literature regarding deprenyl (selegiline). To date, I have read hundreds of published papers about deprenyl. Only about half of these papers were indexed in Medline (the US electronic medical database, upon which most US experts rely). The other half were indexed in Embase (Exerpta Medica, the European electronic medical database). These papers are a primary basis of my scientific knowledge of deprenyl.[6]
These deprenyl papers cover clinical studies, analytical methods, synthesis (relating to production and manufacturing), toxicology, pharmacology, and catabolism (breakdown and excretion). They cover both animal and human research.
In addition to these data, I have also studied a variety of analyses of deprenyl. The most useful technique for deprenyl is high pressure liquid chromatography.[4] To date, I have analyzed HPLC analyses of four different brands of deprenyl.
The assertions of government witnesses that DEDI’s LDC has the same risk profile as Somerset’s Eldepryl is without scientific merit. It would be analogous to FDA scientist stating that different manufacturer’s tryptophans were of equal risk, which we now know to utterly nonsensical in the face of the morbidity and mortality associated with Showa Denko’s contaminated tryptophan.
Like with L-tryptophan, people died from using Eldepryl.[9] Their deaths are recorded and documented in a published scientific study (the largest and longest study ever undertaken) conducted by the Parkinson’s Disease Research Group (PDRG) of the United Kingdom.[5]
My analysis of the PDRG data shows that the entire excess mortality observed in the study occurred in a single year of a five-to-six-year study.[7] This highly improbable occurrence is completely inconsistent with a general toxic influence of deprenyl itself. It is also at odds with every other human (and animal) trial of deprenyl ever published.
This documentation of deaths associated with the use of Eldepryl scientifically undermines any assertion that different brands of deprenyl are essentially the same. The specific circumstances of the Eldepryl-associated deaths makes it clear that an impurity, not deprenyl itself, was responsible for those deaths.[7] This puts lie to any presumption of equivalent public hazard. The PDRG data establishes clearly and unequivocally that different brands of deprenyl are not only different in their HPLC impurity profiles, but that they have been different in their safety, efficacy and side effects.[10] Furthermore, the fact that DEDI’s deprenyl product is not associated with any mortality or morbidity of any kind places a burden upon the Court to reject the government’s assertions of comparable public hazard.
History is replete with examples of scientists who claimed that something was true or impossible, in the face of evidence contradicting their opinions (or beliefs). True scientists always (always!) reevaluate their premises and conclusions when faced with evidence that contradicts their model of the world. Despite this ideal, in the real world, most scientists choose not to reevaluate their premises. There is an emotional cost to questioning beliefs that are closely held. It is far easier (emotionally) to deny the validity of the contrary evidence. There are also political and economic costs associated with challenging the status quo.
When such presumptions and beliefs are challenged within the judicial system, it is not the responsibility of the court to provide aid and comfort to one side of what is supposed to be a balanced adversarial process. Such partiality necessarily decreases the evidentiary burden on one side and increases it on the other. Instead, the court’s primary responsibility is to remain impartial, so that standards of evidence may be equally applied. To this end, each piece of evidence must be evaluated for its truthfulness. Scientific findings must be found to be consistent with scientifically validated principles. In an adversarial process, scientific findings submitted as evidence by one side must survive the scrutiny of scientists on the side, using scientific principles that should be common to both sides. Such dynamic balance is an ideal of US jurisprudence.
I think that that balance has been abrogated by my dismissal. The defense was deprived of its right to present valid scientific evidence that would have undermined the partisan testimony of prosecution witnesses. (The prosecution was also deprived of its chance to cross-examine my testimony.)
My testimony was not about the structure of deprenyl. The structure of deprenyl was not an adversarial issue in the trial. Everybody knows what it is. That knowledge has reached scientific consensus. Even if you thought that I was not qualified to know what it is because I had not tested it with test tubes, microscopes and “what chemists do,” am I not qualified to know what every scientist thinks it is? And within an adversarial process in which both sides stipulate an issue, is it appropriate for the court to dispute such consensus for one side and not the other?
The adversarial issue of my testimony was different. The prosecution’s position was that LDC and Eldepryl were dangerous to similar degrees. The defense’s position was that they were not, that LDC was fundamentally safe. The structure of deprenyl, and the specific structure of any and/or all deprenyl impurities, have nothing to do with whether or not the impurities do or do not, can or cannot, may or may not, affect safety, or efficacy, or side effects, or any other difference between the two products.
From a scientific perspective, one cannot separate the effects of an impurity from a primary ingredient without physically separating them from each other and measuring their effects separately. Whatever burden of proof the prosecution should have been required to provide to support their contention that deprenyl itself was solely responsible for the public health hazards of deprenyl products was waived by your decision to deny the defense the opportunity to present evidence that impurities can affect public hazard, and in fact, have done so in the past (the PDRG study). You have, de facto, accepted that the primary ingredient (deprenyl) is the sole influence, and that the impurities (because they are not characterized?) are not relevant. Such presumption is scientifically erroneous.
I have examined HPLC analyses of different samples of deprenyl (Eldepryl, Jumex, LDC and Cyprenil). I can say with honesty, certainty and expertise that some of the peaks in the HPLCs are the same and some are different. It is my opinion that every scientist you put in my position would agree with me on that conclusion.
It is also my expert opinion that no scientist would testify that they knew whether or not any particular impurity peak contributed to the side effects, toxicity or hazard of the product, without isolation of that peak and testing it independently. Since this latter step has not been done, by anybody, regardless of possible partisanship to the prosecution or defense, the government’s assertion of equal public health danger cannot be resolved by direct (scientific) means. Therefore, it should not be resolved by any presumption of the Court.
But what is far worse is that the defense’s scientific evidence about the toxicity of unknown deprenyl impurities was suppressed. In other words, the government’s unproven assertion was accepted without evidence, and the defense’s evidence to the contrary was suppressed. This represents the most extreme kind of judicial favoritism.
Does the defense’s evidence stand up to scientific scrutiny? I think it does, based on my training as a scientist, my keen observational and reasoning skills, and its acceptance by other scientists and physicians with comparable scientific qualifications. Unfortunately, the judicial process that would have qualified my evidence within your court has been bypassed. I do not know of a way that can be rectified. But I hope that you can find one.
I have gone to great lengths to present my research in as great a detail and with as much clarity as I can. However, I suspect that I have been unable to anticipate every possible misunderstanding or question, especially given my lack of knowledge of my audience and the non-interactive format of an affidavit. Nevertheless, I have and will continue to welcome any and all dialog about my research and methods.
My research and methods were well known to the prosecution. Not only did I fully disclose the evidence (and my methods) to the prosecution and their scientific experts during the morning before my testimony, but the evidence has been in the public domain for many years (in print and on CERI’s web site).
In a brief face to face meeting with me early this year, Mr. Rubinstein admitted longstanding familiarity with CERI’s web site. Mr. Kimball had previously told me that Mr. Rubinstein had told a Grand Jury that CERI’s web site was owned by Mr. Kimball, so I took that opportunity to question Mr. Rubinstein about his representations and then admonished him for his false statements about CERI. I clearly instructed him to cease and desist stating, intimating or repeating his falsehoods. I hereby reaffirm, for the record, that CERI’s publications and web site are owned solely by CERI, maintained by me personally, and in no way controlled by Mr. Kimball.
Footnote 1. Eighteen grams of water (a little more than a tablespoon) contains approximately 602,300,000,000,000,000,000,000 (6.023 x 10^23) water molecules. A tenth of one percent (one part per thousand) of an impurity would consist of approximately 600,000,000,000,000,000,000 (6 x 10^20) molecules (assuming an equal molecular weight). An impurity of one part per million (ppm) would consist of approximately 600,000,000,000,000,000 (6 x 10^17) molecules. An impurity of one part per billion (ppb) would consist of 600,000,000,000,000 (6 x 10^14) molecules. An impurity of one part per trillion (which is presently difficult to measure on a routine basis) would consist of approximately 600,000,000,000 (6 x 10^11) molecules. So the tiniest of impurities is, in reality, approximately half a trillion molecules per tablespoon of water.
The above numbers are based on an assumption of equal molecular weight between water and impurity. That is not necessarily so. If the impurity is ten times heavier (the approximate weight of a deprenyl molecule), there would be one tenth as many molecules as discussed above. If the impurity was a hundred times larger, there would be a tenth again as many molecules. Regardless, we are still talking about multiple billions of molecules for minor impurities and multiple millions of molecules for trace impurities.
The analytical equipment (HPLC and GC/MS) currently used in sophisticated pharmaceutical laboratories to detect impurities and in medical-testing laboratories for diagnostic purposes routinely fails to detect substances present in less-than-parts-per-billion levels.
How many multi-billion-molecule impurities are present in selegiline, citric acid, water and glycerol? Nobody knows. Can anybody state that billion-molecule impurities are not present in any pharmaceutical, food or dietary-supplement product? No. Can any scientist state that they know “all” the chemicals, substances or ingredients in any product? No. Can they state that they know all the “primary” or “predominant” ingredients? Yes, because that question specifically excludes the impurities. However, a statement that they know all the “significant” or “important” ingredients presumes knowledge of the effect of substances that have not been identified, isolated or tested in any way. Thus, there is no evidence to submit in support of such a conclusion. It is therefore scientifically unfounded.
There are known examples of toxic substances that are active at extremely low doses. For example, aflatoxin is 10,000 times more carcinogenic than alcohol (which is the second most significant cause of cancer in our society, behind tobacco). Snake and insect venoms, dioxin, and MPTP (MPP+) are other examples of extremely potent chemicals that are active at parts-per-billion concentrations. These highly potent chemicals are not limited to toxic moieties; cytokines (cell signaling factors) often operate at nanomolar (10^–9) concentrations. It is scientific hubris to state that we know all such chemicals.
Footnote 2. Some impurities can act as catalysts in the degradation of other ingredients. For example, the traces of copper and iron in highly purified water (or fruit juice) are sufficient to degrade vitamin C in the presence of atmospheric oxygen. Without such impurities, vitamin C and oxygen do not readily react.
Footnote 3. Standardization involves diluting more potent extracts and fortifying less potent extracts so that they are uniform by a specified criteria. For example, the most popular standardized Ginkgo biloba concentrate contains 24% ginkgosides.
Although standardization does make one chemical (or group of chemicals) more standard, it does not necessarily decrease the variability of other ingredients in the product. In fact, it can increase that variability. Thus, standardized products are still unique.
Footnote 4. Chromatography is a isolation and detection technique by which a sample is injected in one end of a long tube filled with a polymer or particulate matrix (the “packing”) through which a liquid solvent or gas flows (like water through sand or dirt, or air through the leaves and branches of a forest of trees). As the sample flows through the matrix, different chemicals in the sample interact with the matrix to different degrees. These differences slow down the passage of the different chemicals to different degrees. At the other end of the tube, the chemicals emerge sequentially, the faster chemicals first and the slower chemicals last. Each time a chemical emerges, it is measured by a detector, which is able to distinguish between the emerging solvent and non-solvent components. The signal from the detector is sent to a graph, which records the emerging chemicals as peaks on a paper graph.
There are many different kinds of chromatographs. Scientists pick the tube diameter, length, packing and solvent (or gas) mixture to optimize the separation of chemicals in their sample. Ideally, every different chemical gets separated. However, it doesn’t necessarily happen that way. Sometimes peaks overlap or coincide. Scientists must take this into account.
In a recent study of 5-hydroxytryptophan (a chemical relative of tryptophan), “peak X” was found to be up to five different chemical substances. Some 5-HTP samples had all five substances, others had only four or three of the five.
Sometimes, impurities have very high affinity for the matrix and have extremely long transit times. If the chramatographic procedure is terminated prematurely, these can be missed. Two such deprenyl impurities have been noted in European HPLCs, but not US HPLCs, because the US standard (USP) specifies a shorter-duration analytical procedure, which is necessarily unable to detect those “slower” impurities.
Footnote 5. The PDRG researchers attributed the deaths solely to the toxicity of deprenyl itself. Yet their conclusion was offered without any supporting data, scientific reasoning, or statistical analysis! Given that their conclusion diametrically contradicted the results and conclusions of a body of pre-existing scientific research, an analysis and discussion should have been provided. The fact that it was not is contrary to scientific traditions, expectations and standards.[11]
Footnote 6. During the trial, you made a comment about book learning being in some way invalid. I was and still am flabbergasted. Such thoughts seem Kafkaesque in their consequences. Books, scientific journals, and scientific meetings are a foundation of scientific expertise. They are also the primary basis of scientific education and training. It has always been that way and it will always be that way.
Footnote 7. Without access to the raw PDRG data, I have been forced to use various mathematical and geometric techniques to analyze the study’s data. These techniques are mathematically and scientifically valid, despite the imprecisions that result from extracting data from a graph.[8] These data and analyses have been published, and have been posted on CERI’s web site for years.
I will now present and discuss the exhibits that I had brought with me to the trial, plus a few additional graphics that I think will clarify aspects of my analysis that I had planned to accomplish verbally at the trial.
Exhibit 1 is the graph from the PDRG study showing 60% increased mortality from deprenyl over the 5-6 year study period (study patients were not recruited simultaneously, so some patients were in the study longer than others). The purple line is the cumulative deaths in the deprenyl group (deprenyl-plus-dopa). The dotted blue line is the deaths from the control group (dopa-only). As you can see, these two lines remain close together until 29 months into the study, whereupon they diverge rapidly for about 6-12 months, and then remain diverged to the same degree for the remainder of the study.
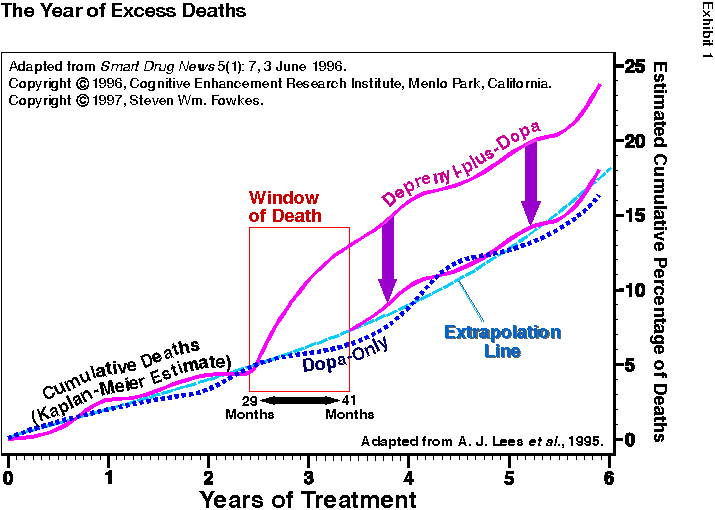
What this means is that the death rate between the deprenyl and control groups were 1) identical during the first 2.4 years of the study, 2) widely different for a period of 6-12 months, and 3) identical for the remaining 2+ years of the study.
To demonstrate this without access to the primary data, I graphically “subtracted” the excess deaths from the 2.4-to-3.4-year period by replacing that segment of the death curve with an “average” (extrapolated) death curve (see blue-dashed line in Exhibit 1). By lowering the purple deprenyl-plus-dopa line down to align with the blue-dashed extrapolation line (follow the purple down-arrows), the excess deaths from the 1-year period (from 29 to 41 months) are removed. The resulting deprenyl-plus-dopa deaths (lower purple line) tracks with the control deaths (blue dotted line) for the remainder of study. This shows, graphically, that the entire increased mortality in this study happened in only one year.
I do not know (and am not qualified to calculate) the statistical unlikelihood of this being a random event. However, I have seen data from thousands of studies similar to this and have never seen anything remotely like this data — except in cases of poisoning or contamination. Considering that this was the largest and longest deprenyl study ever conducted by mankind, it should have far less variation than every other study. Instead, it has ten times more variation. This is not well-behaved data. Something unusual happened during the study.
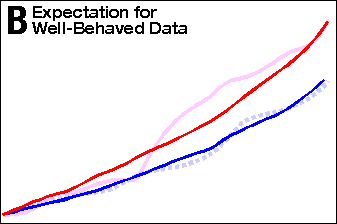 In a well behaved study, the 60% increased mortality would
result in a line of higher slope, that starts out at zero at the
beginning of the study and continues to climb throughout
the study (see Exhibit 2, Panel B). In other words, the two lines
should gradually diverge (move farther apart) as the study
progresses. That is not what happened. The deprenyl group slope
is the same as the control group for the first 2.4 years of the study,
whereupon it suddenly increases dramatically, and then slowly levels
off at its pre-2.4 year slope (see Exhibit 2, Panel C).
In a well behaved study, the 60% increased mortality would
result in a line of higher slope, that starts out at zero at the
beginning of the study and continues to climb throughout
the study (see Exhibit 2, Panel B). In other words, the two lines
should gradually diverge (move farther apart) as the study
progresses. That is not what happened. The deprenyl group slope
is the same as the control group for the first 2.4 years of the study,
whereupon it suddenly increases dramatically, and then slowly levels
off at its pre-2.4 year slope (see Exhibit 2, Panel C). 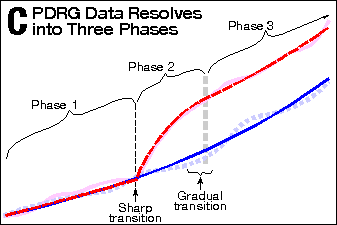 During Phase 1, the deaths
are matched. During Phase 2, they diverge dramatically. During
Phase 3, they are again matched (parallel). Please note that the
transition from Phase 1 to Phase 2 is abrupt. I will get back to
the significance of that observation later.
During Phase 1, the deaths
are matched. During Phase 2, they diverge dramatically. During
Phase 3, they are again matched (parallel). Please note that the
transition from Phase 1 to Phase 2 is abrupt. I will get back to
the significance of that observation later.
The PDRG researchers advanced the hypothesis that the increased mortality resulted from a delayed toxic effect of deprenyl. Is this view valid? Does it survive scientific scrutiny?
The biggest inconsistency is the death rate returning to normal after a year of toxicity. If we were merely dealing with delayed toxicity, the death rate should have stayed higher. It did not. This means that we must be seeing an example of a temporary delayed toxic effect. Such a hypothetical effect has never been observed before and should, therefore, be accepted only with strong evidence and extensive analysis. The PDRG researchers provided no such analysis, whatsoever.
The second inconsistency has more to do with wisdom. When there are two hypotheses that explain data, one must consider the relationship between those explanations and the rest of scientific knowledge. For example, has this toxic effect of deprenyl ever been observed in other human studies? The answer is no. However, the other human studies of deprenyl were only one or two years in length, which is insufficient to observe a toxic effect that suddenly starts at 2.4 years.
What about animal studies? There have been many lifespan studies of deprenyl in rodents, none of which have shown any evidence of a delayed toxic effect. Quite the opposite effect is routinely seen — an extension of lifespan that extends to extreme ages.
The third inconsistency is the abruptness of the delayed toxic effect. Given the wide variations in age, metabolism, immunity and genetics of Parkinson’s patients enrolled in the study, a time-delayed toxicity would not be expected to be abrupt — at all. The PDRG data is strikingly abrupt. On the other hand, abruptness is a defining characteristic of cases of contamination (poisonings).
Is there an alternative hypotheses to a “delayed toxic effect?” Certainly. It could be toxicity from impurities or contamination.[12] There are many examples of sudden increases in mortality (or morbidity) from impurity and contamination incidents, which quickly resolve when the toxic agent is recognized and eliminated. Cyanide in Tylenol, Showa Denko tryptophan, toxic oil syndrome, bad cheese, cases of common food poisoning, and many more. The “temporariness” of such toxic events is inherent in the process. As such, it requires no new hypothesis, circumvention of logic, or voodoo science to explain.
The Showa Denko-tryptophan incident is a good example of an impurity/contamination event. For twenty years, people used tryptophan safely as a dietary supplement. Suddenly, in 1989, hundreds of people started getting sick and dozens died (i.e., the mortality and morbidity rates suddenly spiked). When Showa Denko’s tryptophan was removed from the market, people stopped getting sick and dying (i.e., the mortality and morbidity returned to normal).[9]
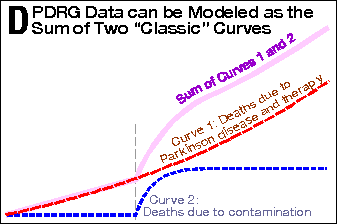 Getting back to Exhibit 2, the PDRG data can be modeled as the
sum of two separate influences (see Panel D). The first influence
is Parkinson’s disease itself, which shows a smooth death
curve (red dashed line) which increases in slope over time.
This is classic for neurodegenerative diseases. The second
influence is a contamination (poisoning)
event,[12] whose curve
(dashed blue line) suddenly (abruptly) increases at a specific point
in time, and levels off after the contamination is eliminated and
its effects wear off. Again, this is a classic curve for that kind
of phenomenon. When these two influences are added together, the
produce a line (pale magenta) which closely reproduces the PDRG data.
Getting back to Exhibit 2, the PDRG data can be modeled as the
sum of two separate influences (see Panel D). The first influence
is Parkinson’s disease itself, which shows a smooth death
curve (red dashed line) which increases in slope over time.
This is classic for neurodegenerative diseases. The second
influence is a contamination (poisoning)
event,[12] whose curve
(dashed blue line) suddenly (abruptly) increases at a specific point
in time, and levels off after the contamination is eliminated and
its effects wear off. Again, this is a classic curve for that kind
of phenomenon. When these two influences are added together, the
produce a line (pale magenta) which closely reproduces the PDRG data.
Two years after I performed this analysis, I was asked to revisit the PDRG data by a subscriber. In the process of reconsidering my previous explanation, I realized that some people have a hard time interpreting cumulative death curves. I realized that they needed to see the data as a death-rate curve (deaths per unit time). Without the primary data, I was again forced to use geometric means to accomplish this end.[8]
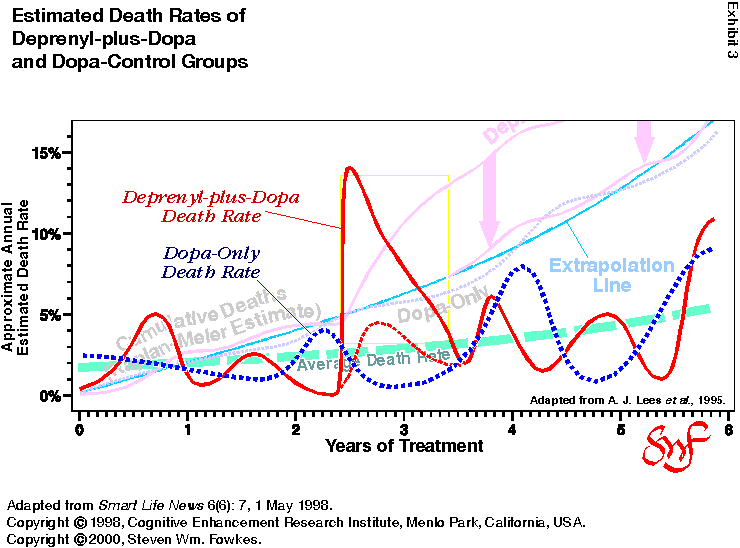
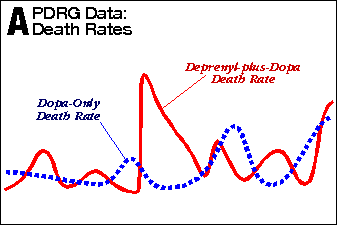 Exhibit 3 shows the PDRG data as a death-rate curve. (I’ve
left in pale images of the cumulative death curves so that you
can compare slopes if you wish.) As you can plainly see,
the spike in death rate in the deprenyl-plus-dopa group (solid
red line) at 2.4 years is highly unusual. It is not only different
in magnitude, it is different in shape. The abruptness of the increase
(going from less than 1% to 14% in one month is quite consistent with
a contamination event and completely inconsistent with a “delayed
toxic effect.”
Exhibit 3 shows the PDRG data as a death-rate curve. (I’ve
left in pale images of the cumulative death curves so that you
can compare slopes if you wish.) As you can plainly see,
the spike in death rate in the deprenyl-plus-dopa group (solid
red line) at 2.4 years is highly unusual. It is not only different
in magnitude, it is different in shape. The abruptness of the increase
(going from less than 1% to 14% in one month is quite consistent with
a contamination event and completely inconsistent with a “delayed
toxic effect.”
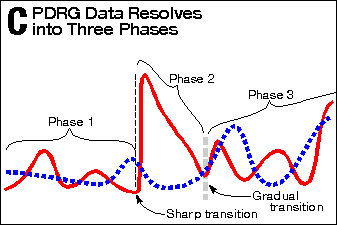
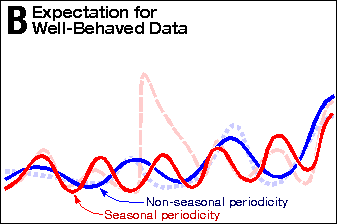 Exhibit 4 shows a similar analysis of Exhibit 3 as Exhibit 2 did of
Exhibit 1. Panel B shows what well behaved data might look like.
(I have no explanation for why the dopa-only
death rate does not show the almost perfect seasonal variation that
the dopa-plus-deprenyl group does.) In Panel C, the same three phases
can be seen.
Again, the Phase-1-to-Phase-2 transition is exceedingly abrupt,
while the Phase-2-to-Phase-3 transition is gradual. In Panel D,
the PDRG death-rate curve is modeled as the sum of two curves. The
death-rate curve from Parkinson’s disease (dashed red line),
added to the death-rate curve from contamination (the dashed blue
line), produces the PDRG death-rate curve (pale magenta solid line).
Exhibit 4 shows a similar analysis of Exhibit 3 as Exhibit 2 did of
Exhibit 1. Panel B shows what well behaved data might look like.
(I have no explanation for why the dopa-only
death rate does not show the almost perfect seasonal variation that
the dopa-plus-deprenyl group does.) In Panel C, the same three phases
can be seen.
Again, the Phase-1-to-Phase-2 transition is exceedingly abrupt,
while the Phase-2-to-Phase-3 transition is gradual. In Panel D,
the PDRG death-rate curve is modeled as the sum of two curves. The
death-rate curve from Parkinson’s disease (dashed red line),
added to the death-rate curve from contamination (the dashed blue
line), produces the PDRG death-rate curve (pale magenta solid line).
This analysis shows that an impurity (contaminant)
in Eldepryl was fully responsible for the increased mortality seen
in the PDRG study.[12,13] The corollary is that deprenyl
(selegiline) is not the causal influence in those deaths. This
provides clear scientific evidence that public safety considerations
must address the risks associated with specific impurity ingredients
found in deprenyl products. The fact that this issue is not being
investigated by governmental agencies with public-safety responsibilities
does not invalidate its relevance to the Kimball case. The fact
that the toxic agent(s) remain unknown does not justify 1) blaming
deprenyl for that hazard, or 2) assigning the demonstrated dangers
of Eldepryl to LDC.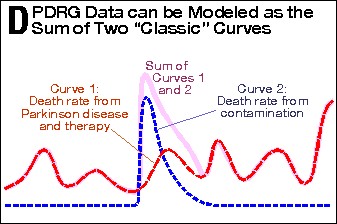
Footnote 8. In calculus terms, death rate is the first derivative of cumulative deaths. In non-calculus terminology, the death rate at any point in the study is equal to the slope of the death curve at that same point.
An analogy might make this clearer. If you were driving from one city to another, the distance driven could be plotted over time. This would be a distance curve. Each point on the curve would be the number of miles you had driven at that point in time. The slope of the distance curve would be your speed, how many miles per hour you were driving, at that point in time. Speed is the change in position per unit time.
If, during the trip, you stopped for lunch, you would not be getting any closer to your destination during the break. The distance curve would be perfectly flat, and the slope during that time would be zero, which is your rate of speed while sitting in a restaurant.
In calculus, these curves can be expressed as differential equations. The speed equation is the first derivative of the position equation. The second derivative would be acceleration equation (the change in speed per unit time, or the change in change of position per unit time).
Derivatives of differential equations have a simple geometric relationship to each other. That relationship is slope. The slope of a curve at any point is always equal to the value of the derivative at that same point. Just as the slope of the position curve is equal to the speed, and the slope of the speed curve is the acceleration, the slope of the PDRG death curve is the rate of death.
Mathematically, there is no essential difference in validity between using calculus or geometry in producing a derivative curve.
Footnote 9. Interestingly, the FDA still officially blames tryptophan for the deaths caused by the contaminant. Like the PDRG scientists, FDA scientists have employed flawed procedures and illogical arguments to support their politically preconceived conclusions. If you wish, I can present evidence of 1) scientific fraud and 2) corruption of the peer-review process, in a case of FDA-funded and FDA-supervised tryptophan research.
Footnote 10. Although I spend considerable time and effort discussing the PDRG study, that is not the only available evidence indicating a difference between different deprenyl products. I have received hundreds of anecdotal reports over the last eight years of dramatic differences in subjective and clinical responses to different brands of deprenyl. Many of these anecdotes involve patients switching from one kind of deprenyl to another. Annetta Freeman’s experiences are but one example. More than a dozen have come from medical professionals. All such reports of differences assign the adverse side effects to Eldepryl and Jumex. I have yet to receive a single report of increased side effects from LDC.
I do not dwell on these reports because they are anecdotal. However, they do deserve mention. They are consistent with my analysis of the PDRG data. They also support Mr. Kimball’s claim that LDC is much safer and more efficacious than Eldepryl.
These reports include my own experiences. On one occasion, I undertook a personal experiment with deprenyl. On a workday morning, I took a 5 mg Jumex tablet, which is ten times my normal dose of one mg LDC every other day. I experienced a spectrum of powerful anti-cognitive effects which I can only describe as debilitating and unpleasant. I was completely unable to write effectively. I was also unable to sleep well that night, despite taking two grams of L-tryptophan with melatonin. The was a persistent residual anti-cognitive effect the next day.
A week later, I took 5 mg of LDC. Although I became excessively stimulated by that dose (far more than from the Jumex the week before), I experienced none of the anti-cognitive effects that I had from Jumex. My writing was extremely productive. Even though I was not able to relax effectively at the end of the day, the experience was pleasant overall. I was able to sleep through the night with the aid of tryptophan and melatonin.
My experiences, and those of Dr. Ward Dean, were written up and published in Smart Drug News 5(1): 3 June 1996. That article is also posted on our web site (www.ceri.com/depren.htm).
Footnote 11. Dr. Lees of the PDRG has failed to respond to my letter requesting access to the study’s raw data, from which I could commission an independent statistical analysis.Footnote 12. Although I am ethically prohibited from disclosing the nature of the proprietary manufacturing process developed by DEDI, I do have reason to believe that it would effectively remove a class of impurities known as quaternary ammonium salts. Unlike amphetamine, methamphetamine, desmethylselegiline and selegiline, quaternary ammonium salts always have a positive charge (see Exhibit 5). This is because the nitrogen atom (N) has four carbon-based molecular groups attached to it, instead of three (for deprenyl), two (for methamphetamine and desmethylselegiline) or one (for amphetamine).
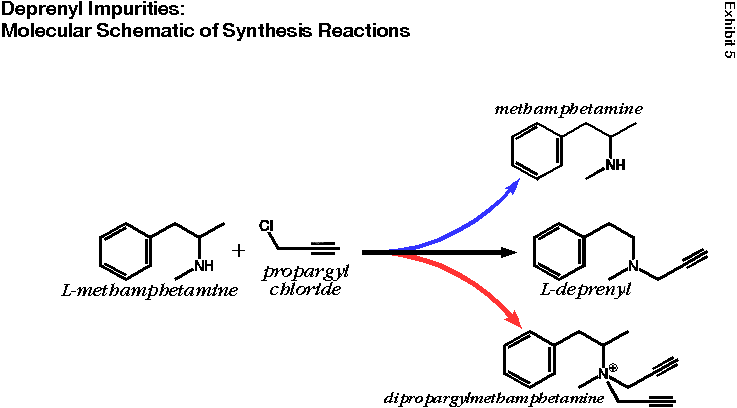
One specific quaternary ammonium salt is an expected side reaction of the standard manufacturing process utilizing L-methamphetamine and propargyl chloride (see Exhibit 5). During alkylation reactions (reactions where carbon-based groups are “attached” to another molecule), the molecules do not necessarily react in a one-to-one manner. Remember, a liter-sized deprenyl synthesis reaction involves hundreds of thousands of millions of billions of methamphetamine molecules reacting with hundreds of thousands of millions of billions of propargyl chloride molecules. As you might expect, most methamphetamines will get one propargyl group (black arrow), but some will get none (blue arrow) and some two (red arrow). Dipropargylmethamphetamine is therefore an expected reaction byproduct for this method of deprenyl synthesis.
The reason I am concerned about quaternary ammonium derivatives of deprenyl is that they are structurally related to a class of known poisons (see Exhibit 6). These poisons are of specific interest because they selectively interfere with the activity of coenzyme 1 (NAD), which helps create biological energy at the cellular level. NAD helps generate energy by binding to Complex I, which is located within the many tiny mitochondria (power plants) of the body’s cells. This is the first step in a chain of events that produces 90% of the biological energy we need to stay alive.
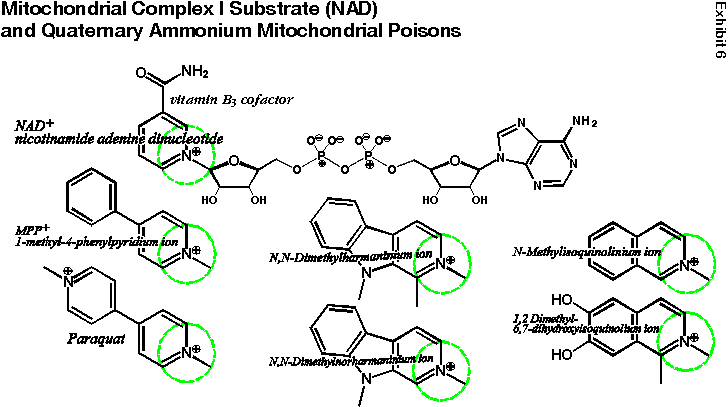
Complex I is known to be selectively impaired in Parkinson’s disease. One of these mitochondrial poisons (MPP+) is known to be able to induce permanent symptoms of Parkinson’s disease in both animals and people in just 24 hours!
Complex I poisons share a common structural feature with NAD: they have a quaternary (positively charged) nitrogen atom (see dashed green circles) attached to (or next to) an aromatic ring system (the hexagons with the three extra lines inside). Quaternary deprenyl derivatives have a quaternary ammonium nitrogen next to an aromatic ring system.
Quaternary ammonium deprenyl derivatives may not be produced solely during the synthesis of deprenyl. Disproportionation reactions can produce two different deprenyl quaternary derivatives (see Exhibit 7). Disproportionation is a chemical term that means deviation from an ideal one-to-one molecular ratio. In this process, two deprenyl molecules react with each other to transfer a carbon group from one to the other. If they transfer a methyl group (see red pathway 1), dimethylpropargylamphetamine results. This has the positively charged nitrogen next to the aromatic ring (see dashed green circle). If they transfer a propargyl group (see blue pathway 2), dipropargylmethamphetamine results (the same quaternary ammonium derivative mentioned earlier).
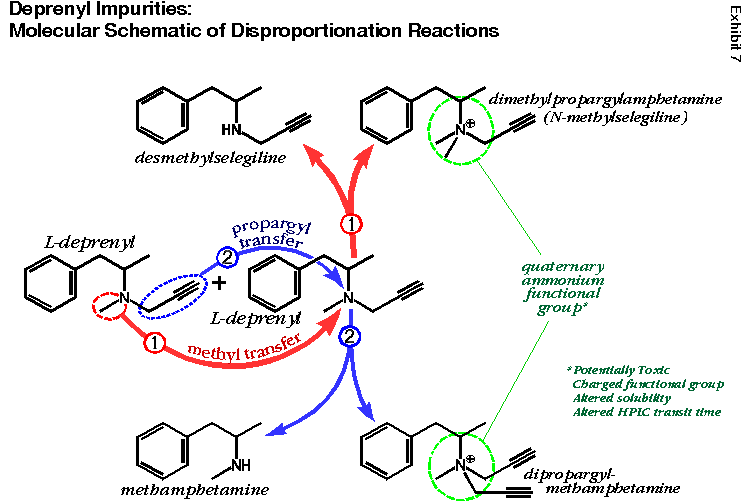
Due to their positive charges, quaternary ammonium compounds tend to have long retention times in HPLCs that are optimized for amines (i.e., deprenyl and its catabolites). They are not “picked up” by the USP chromatography standard established for deprenyl.
Footnote 13. Interestingly, at the same time as the contamination event or shortly following it, Somerset Pharmaceuticals (the US manufacturer of Eldepryl) stopped importing Eldepryl from Eastern Europe and started manufacturing it in Puerto Rico. Was the contamination event the incentive for this change?

Steven Wm. Fowkes
Exhibit Name |
Exhibit Description |
|---|---|
Exhibit #1 |
The Year of Excess Deaths |
Exhibit #2 |
Cumulative Deaths Analysis A-D |
Panel A |
PDRG Data: Cumulative Deaths |
Panel B | Expectation for Well-Behaved Data |
Panel C |
PDRG Data Resolves into Three Phases |
Panel D |
PDRG Data can be Modeled as the Sum of Two “Classic” Curves |
Exhibit #3 |
Estimated Death Rates of Deprenyl-plus-Dopa and Dopa-Control Groups |
Exhibit #4: |
Death Rate Analysis A-D |
Panel A |
PDRG Data: Death Rates |
Panel B |
Expectation for Well Behaved Data |
Panel C |
PDRG Data Resolves into Three Phases |
Panel D |
PDRG Data can be Modeled as the Sum of Two “Classic” Curves |
Exhibit #5 |
Deprenyl Impurities: Molecular Schematic of Syntheses Reactions |
Exhibit #6 |
Mitochondrial Complex-I Substrate (NAD) and Quaternary Ammonium Mitochondrial Poisons |
Exhibit #7 |
Deprenyl Impurities: Molecular Schematic of Disproportionation Reactions |
------------------------------------------------------------------ Steven Wm. Fowkes (fowkes@ceri.com) Phone: 650-321-CERI Cognitive Enhancement Research Institute Fax: 650-323-3864 P.O.Box 4029, Menlo Park, California, 94026 USA Alt: 650-321-6670 ---------------------http://www.ceri.com/-------------------------